Bionime Continuous Glucose Monitoring (CGM) System Set for Pivotal Trial
Taichung, Taiwan – Bionime, a designer and manufacturer of blood glucose monitoring devices, is in the process of developing its RIGHTEST Continuous Glucose Monitoring (CGM) System. Aimed at improving the lives of people with diabetes, the CGM system provides 24-hour glucose tracking.
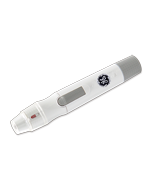
The RIGHTEST CGM provides glucose level measurements at 5-minute intervals for 14 days continuously, eliminating the need to use fingersticks. Additionally, the CGM is capable of delivering advance alerts of impending hypoglycemia or hyperglycemia.
Users can self-apply the sensor to their arm (or abdomen for pediatrics) and connect the rechargeable transmitter. This communicates with a receiver or smartphone, as well as Bionime’s DMS cloud, for data visualization and alerts.
“Bionime strives to help people manage glucose effectively”, says Roy Huang, CEO of Bionime. “This CGM is another technological milestone for us.”
The RIGHTEST CGM promises ease-of-use, as well as a mobile app available for communicating, via the cloud, with hospital systems such as the HIS, allowing triage & continuous two-way interactions.
To learn more about Bionime, visit www.bionime.com
About Bionime
Established in 2003, with the goal of improving lives of people with diabetes through better self-monitoring, Bionime is a leading designer and manufacturer of blood glucose monitoring systems. Accurate, affordable testing is provided through a lineup of blood glucose meters designed for self-monitoring and clinical use, along with patented test strip technology. Headquartered in Taiwan, Bionime products have a global presence through partnerships with distributors worldwide.
Follow Bionime on Facebook or LinkedIn.